1 – Implementation of the quality approach in the laboratory
Like many other research laboratories, Cermav was challenged in 2002 with several questions: retirements, oral transmission of knowledge …, associated with a large range of equipments (electron microscopy, mass spectrometry, NMR, chromatography , X-rays). It was a question of formalizing and memorizing laboratory practices. With this in mind, the ISO 9001 quality management standard (generalist, flexible and internationally recognized) has established itself as the benchmark of choice. A Quality approach was implemented in 2003 across all of our activities, both technical and administrative, in order to meet the requirements of the standard. We were initially interested in quality in research, since it is commonly accepted that only 20% of research activity is devoted to a creative activity, the rest corresponding to routine and classic tasks. The idea was to organize research activities more efficiently by working on the organizational aspects of research.
2 – Aspects of the ISO 9001 standard
Before the practical realization, it is necessary to identify the three aspects of the Quality approach which are the process approach, continuous improvement and documentary control, through their adaptation to the problem of a research laboratory.
2.1 The process approach
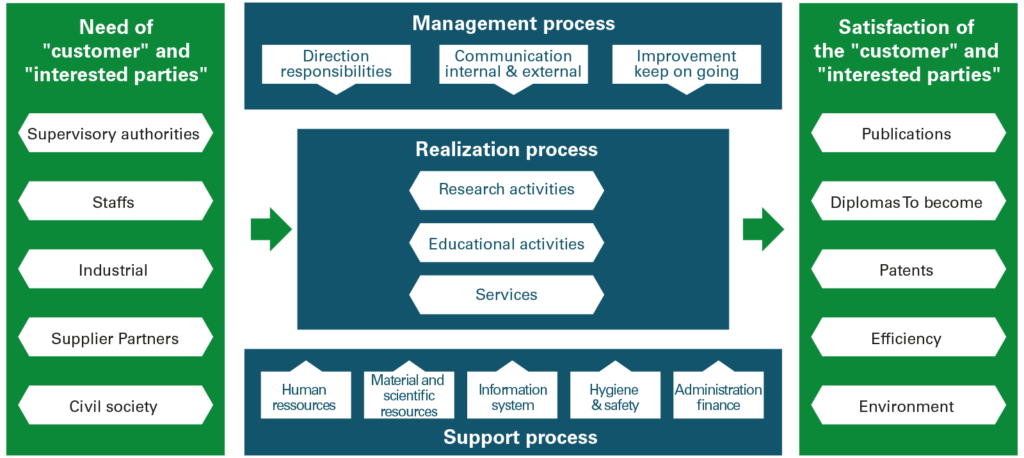
The process approach, which corresponds to the structuring of the laboratory’s activities, consisted in breaking down its functioning into a certain number of processes or major activities. The identified processes (fig. 1) are divided into management processes, support processes and production processes which represent the “core business” processes of the Unit.
ISO 9001 introduces the notion of customer, whose satisfaction will be measured by means of indicators. For example, for our supervisory authority, the CNRS, our scientific output (number of publications, average impact factor, etc.) represents an indicator of the “Research activities” process.
2.2 Continuous improvement
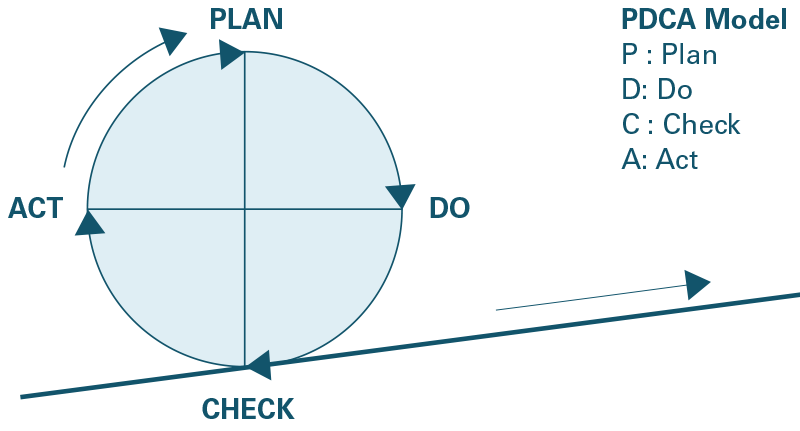
The management system must seek to improve and to do so, tackle all anomalies and dysfunctions that may occur. This principle of continuous improvement is generally represented by a series of corrective and preventive actions symbolized by a “Deming wheel” or PDCA model for Plan / Do / Check / Act (fig. 2).
The successive sequence of these steps will make it possible to go towards an optimization of the system. At Cermav, continuous improvement is essentially based on two tools which correspond to the requirements of the standard: the implementation of internal audits, and management reviews.
2.3 Documentary control
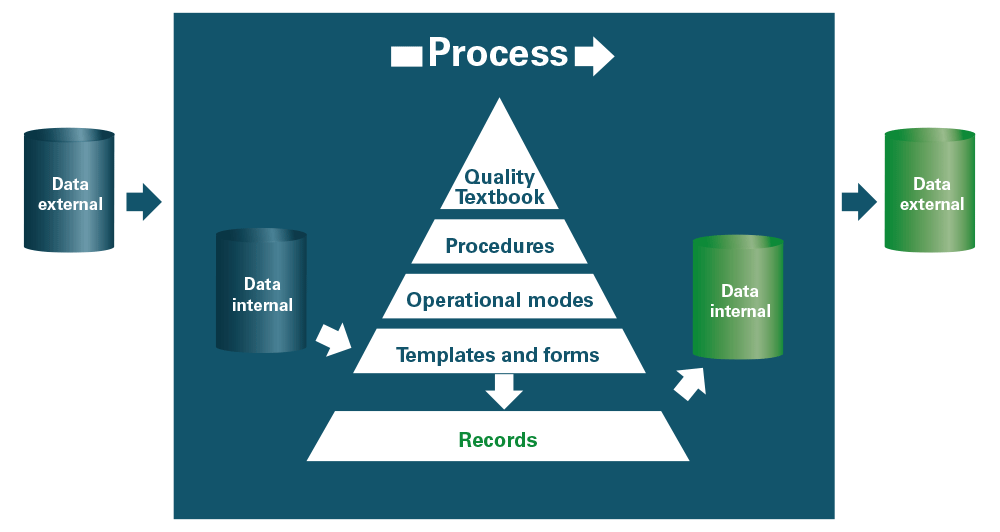
The process approach and continuous improvement are driven by documentary control, which allows information to be structured (fig. 3). The documentary system associates within a hierarchical structure, represented in the form of a pyramid:
- the quality manual, the entry point of the system which describes the processes,
- the procedures which detail the process control provisions (“Who does what?”),
- the operating modes which specify in detail the steps of the procedures (“How?”),
- templates and forms, used to create documents or as recording media,
- the records which provide proof of what has been done and ensure the traceability of the system.
3 – The laboratory organization system
The originality of the approach carried out at Cermav was to combine the redesign of the laboratory’s intranet site and the Quality approach, in a sort of web portal, providing a front-end for access to the laboratory’s internal documentary and application resources. SOURCE is the Organization System of the CErmav Research Unit (fig. 4). The web interface provides access to all the information, whether it is Quality documentation (procedures, operating modes, etc.) or dynamic information, data and records, such as:
- operating procedures for using equipment
- reports (laboratory advice, commissions, etc.)
- reservation schedules (rooms, equipment, etc.)
- access to administrative documents (missions, training, etc.)
- access to databases (personal, publications, etc.)
- access to a collaborative space for the different research teams, monitoring of laboratory notebooks …
The SOURCE tool was selected as part of a call for projects from the CNRS Mission for Technological Resources and Skills (MRCT). SOURCE is available on the site of the Quality in Research network (IT Tools section).

4 – The benefits of the Quality approach
The Source tool, used on the intranet home page, is now integrated into the Unit’s operating mode. Appropriation of the system by the staff has been facilitated by the integration of tools such as diaries, reservation schedules, bibliographic tools … Along with the improvement of the organization and internal information circuits, the The establishment of a quality management system also had an impact on the level of staff involvement and the governance of the laboratory. The intranet and quality approach association fully met the objectives set, by contributing to the improvement of general operations, and by providing clarity to our partners.
5 – Quality of research
Let us now return to the core business of a research laboratory with the quality of the research itself. Indeed, how can one imagine being at the heart of the production of scientific results, without ensuring the implementation of optimal means to guarantee the quality and in particular the traceability of the results produced? At CERMAV, this is ensured by the use of laboratory notebooks and the establishment of project sheets. These project sheets ensure continuity between a scientific project, its description (human and financial resources, etc.), its operation (reports, reports, etc.), the laboratory notebooks used as well as the digital archives including the structuring obeys an operating mode.
It is this same concern to ensure traceability of research activities that has led the laboratory to define, for example, a roadmap for doctoral students including a list of objectives to be achieved during their thesis (fig. 5). and whose regular validation by the thesis supervisor is necessary for registration at the Doctoral School.